30 Jul


Posted By
14 Comment(s)
1001 View(s)
The clinical application of medical endoscopes is a detection instrument that integrates traditional optics, ergonomics, precision machinery, modern electronics, mathematics, and software. It consists of a endoscope system, an image display system, an illumination system, and peripheral equipment, with core components such as image sensors, optical lenses, light source illumination, and mechanical devices. Composition of Endoscopic System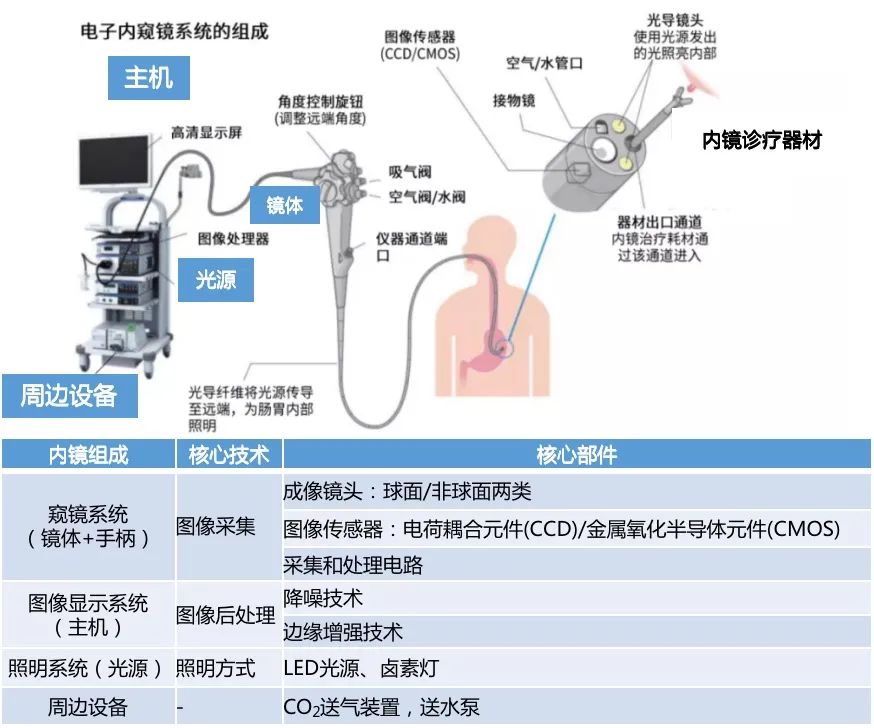
Data source: Olympus official website, Integrated Research
Endoscope is one of the key surgical instruments for achieving minimally invasive diagnosis and treatment. It can enter the body through natural channels or through the skin. When used, the endoscope is introduced into the pre observed organ and directly observes changes in the relevant area. Endoscopes have a wide range of application scenarios and can achieve various functions such as examination, disease diagnosis, and minimally invasive treatment. Depending on the characteristics of different endoscopic products, they can be applied to different departments. Application scenarios of endoscopes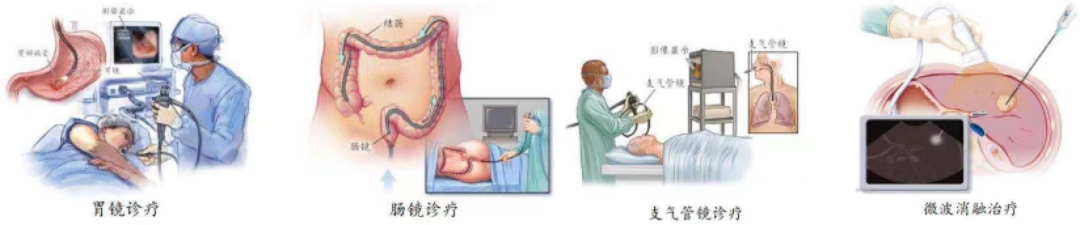
 Data source: Surgery, WHO ICD, Integrated research
Data source: Surgery, WHO ICD, Integrated research
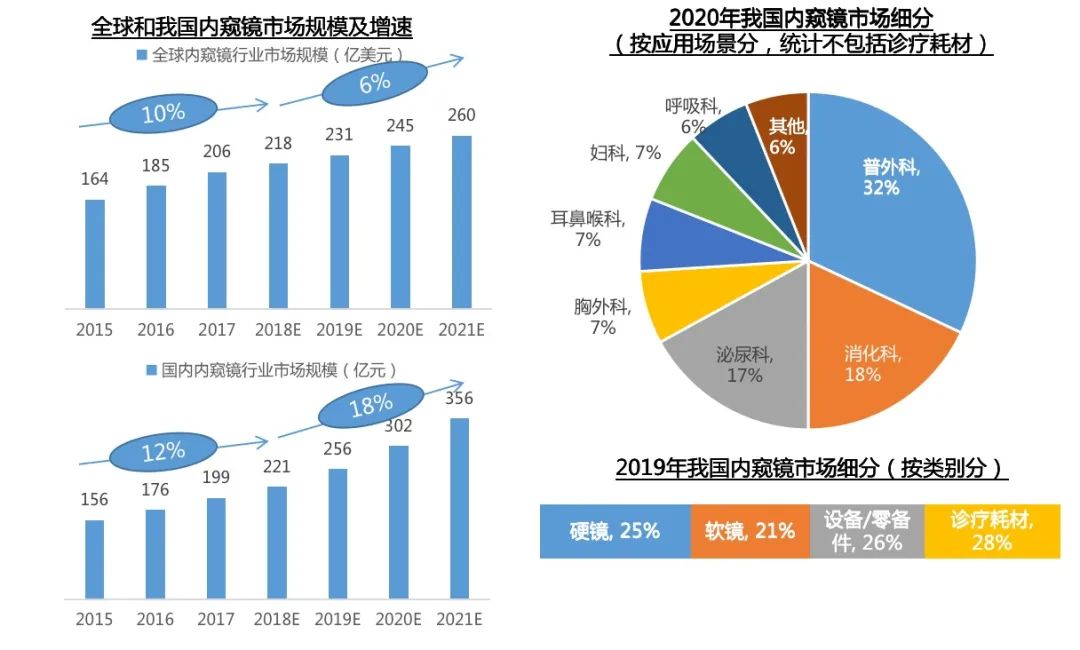
Endoscopes are divided into hard and soft types. Hard mirrors are mainly used to enter sterile tissues in the human body, such as laparoscopy, arthroscopy, etc., with relatively low technical barriers. Soft mirrors can bend freely and mainly enter through the natural cavity of the human body, such as gastrointestinal endoscopy, bronchoscopy, etc. The technical barrier is high, which is reflected in the composite requirements for micro imaging module technology, endoscopic image processing technology, precision structural design and processing technology. The global market size of medical endoscopes reached 23.1 billion US dollars in 2019, with a CAGR of about 6% in recent years; In 2019, the market size of medical endoscopes in China reached 25.6 billion yuan. With the popularization of early screening and diagnosis, and the increasing penetration rate of minimally invasive surgery, the market size continues to expand, with a CAGR of about 18% from 2018 to 2021.
Data source: EvaluateMedTech, Frost&Sullivan, Fusion Research
In 2019, the size of China's hard mirror market was 6.5 billion yuan, with German, Japanese, and American companies occupying the majority of the market share. Domestic brands are mainly concentrated in the mid to low end market, with a localization rate of less than 20%. The current situation of the soft mirror market is completely different from that of the hard mirror market. Due to the monopoly of optical imaging technology, 93% of the global soft mirror market is occupied by the three giants of Japanese companies - Olympus, Pentax, and Fuji. The domestic market also shows a similar trend: in 2019, the size of China's soft mirror market was about 5.3 billion yuan, and the market share of the three giants of Japanese companies was as high as 95%.
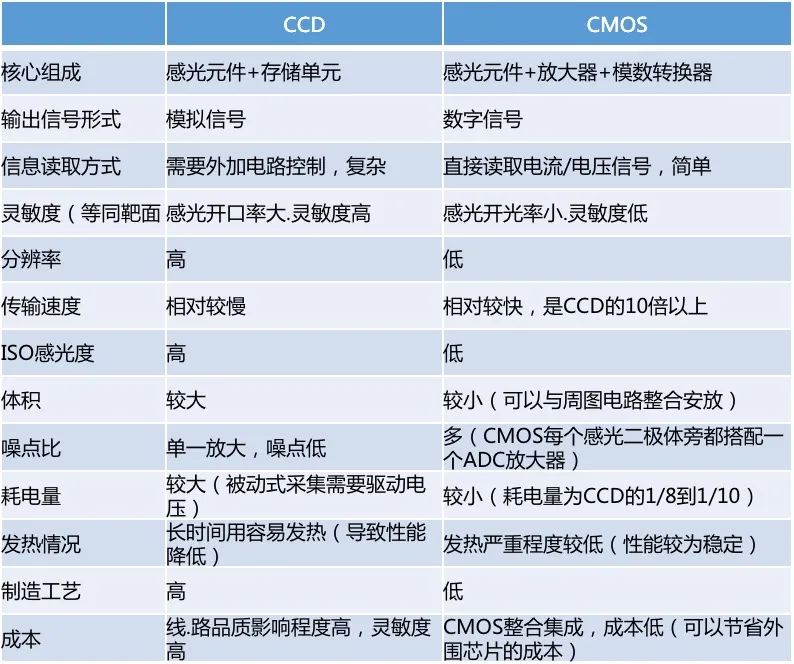
Technological iteration catalyzed commercialization of disposable endoscopic image sensors is one of the core components of soft endoscopes, mainly including charge coupled devices (CCD) and metal oxide semiconductor devices (CMOS). CCD technology has the advantages of high resolution, wide dynamic range, low distortion, high sensitivity, etc. Mainstream endoscopes use CCD technology, but the price is high. Japanese companies have formed a monopoly in the era of electronic soft mirror CCD, and can only obtain low-end products domestically. With the technological upgrade in the civilian mobile phone field, the imaging level of CMOS technology has approached that of CCD, breaking the absolute monopoly of CCD in the field of image sensors. Comparison of CCD and CMOS Technologies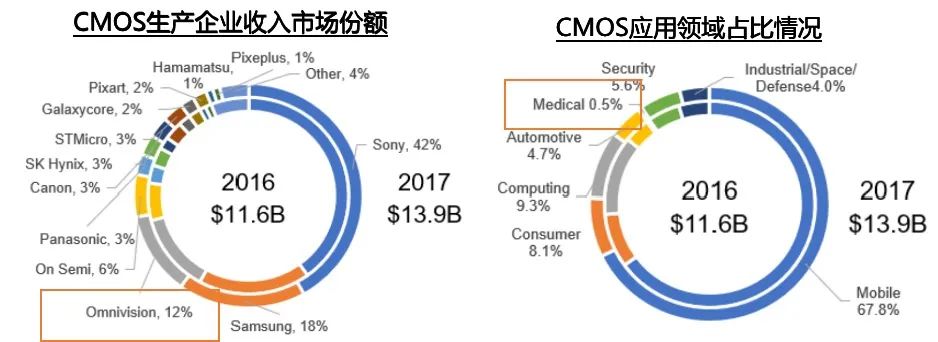

In 2019, the size of China's hard mirror market was 6.5 billion yuan, with German, Japanese, and American companies occupying the majority of the market share. Domestic brands are mainly concentrated in the mid to low end market, with a localization rate of less than 20%. The current situation of the soft mirror market is completely different from that of the hard mirror market. Due to the monopoly of optical imaging technology, 93% of the global soft mirror market is occupied by the three giants of Japanese companies - Olympus, Pentax, and Fuji. The domestic market also shows a similar trend: in 2019, the size of China's soft mirror market was about 5.3 billion yuan, and the market share of the three giants of Japanese companies was as high as 95%.
Technological iteration catalyzed commercialization of disposable endoscopic image sensors is one of the core components of soft endoscopes, mainly including charge coupled devices (CCD) and metal oxide semiconductor devices (CMOS). CCD technology has the advantages of high resolution, wide dynamic range, low distortion, high sensitivity, etc. Mainstream endoscopes use CCD technology, but the price is high. Japanese companies have formed a monopoly in the era of electronic soft mirror CCD, and can only obtain low-end products domestically. With the technological upgrade in the civilian mobile phone field, the imaging level of CMOS technology has approached that of CCD, breaking the absolute monopoly of CCD in the field of image sensors. Comparison of CCD and CMOS Technologies

Data source: CNKI, Integrated Research
CMOS is currently widely used in the fields of mobile devices and cameras, with a small but rapidly growing proportion in the medical industry. Omnivision, a representative manufacturer with a market share of about 12%, was acquired by a domestic enterprise in 2016, accelerating the iteration of optical imaging technology for domestic manufacturers. Haowei has been deeply involved in the medical market for many years, providing endoscope manufacturers with a series of solutions including image sensors, wired modules, ISP boards, and complete reference design solutions。 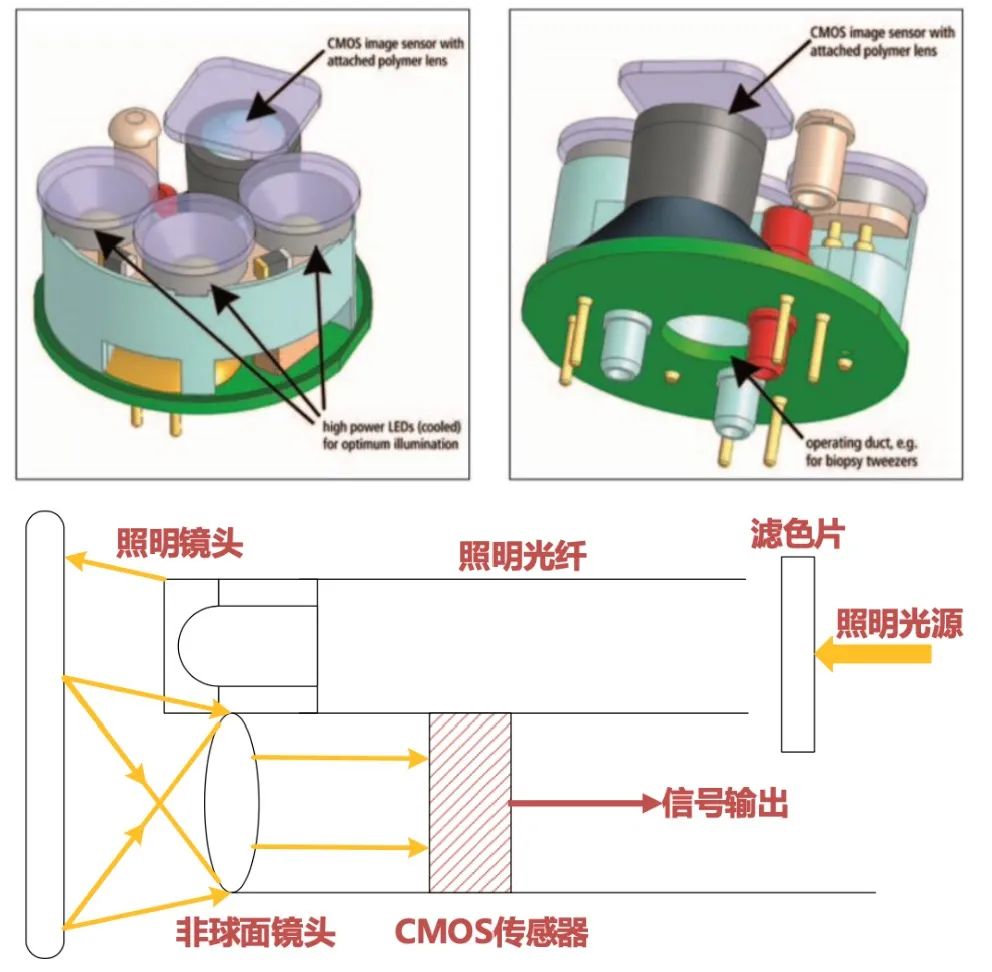 数据来源:融汇Researching the opening of domestic medical brands and the use of CMOS technology to replace CCD by Shanghai Aohua, leading the way in breaking through the soft mirror market. With sufficient supply of high-end CMOS technology and obvious technological advantages, domestic brands can gradually overcome the limitations of CCD and narrow the gap with imported brands. At the same time, the small size and low cost of CMOS chips also make it possible for endoscopes to be consumables, which not only solves clinical pain points but also has greater health and economic value. Structure and Imaging Principle of CMOS Endoscope
数据来源:融汇Researching the opening of domestic medical brands and the use of CMOS technology to replace CCD by Shanghai Aohua, leading the way in breaking through the soft mirror market. With sufficient supply of high-end CMOS technology and obvious technological advantages, domestic brands can gradually overcome the limitations of CCD and narrow the gap with imported brands. At the same time, the small size and low cost of CMOS chips also make it possible for endoscopes to be consumables, which not only solves clinical pain points but also has greater health and economic value. Structure and Imaging Principle of CMOS Endoscope
Data source: Haowei Technology official website, Integrated Research
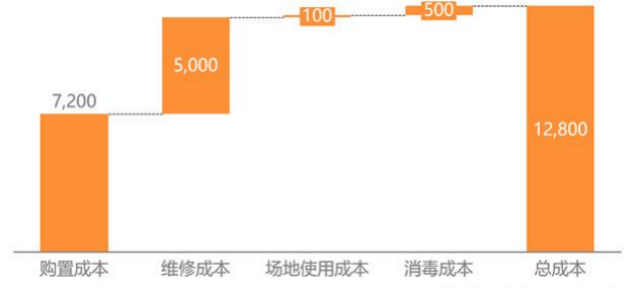
Disposable endoscopes can solve the clinical pain points of cross infection during the use of medical endoscopes. Endoscope is a medical device that comes into close contact with the patient's skin, mucous membranes, and sterile tissues. However, due to the presence of multiple small and long open channels in the endoscope structure, it provides an environment for residual microorganisms, secretions, and blood, as well as cross infection. Medical accidents caused by cross infection often occur. Under the COVID-19 epidemic, the problem of cross infection has attracted great attention. The novel coronavirus can bind to the angiotensin converting enzyme 2 (ACE2) receptor, which exists in the epithelial cells of the lung, upper esophagus, ileum and colon, meaning that the respiratory system and digestive system can bind to the virus. The expert consensus released by the Chinese Medical Association during the epidemic mentioned the use of disposable endoscopes.  Disposable endoscopes have greater health and economic value. A study pointed out that the cost of a single operation of domestic traditional flexible ureteroscope was more than 12000 yuan, and the cost of daily disinfection and sterilization and operation and maintenance accounted for 43% of the total cost. Disposable endoscopes do not need to consider lifespan or maintainability, resulting in reduced usage costs. Taking into account the equipment and consumables, the initial cost of performing surgery with a disposable ureteroscope is 5% of that of a reusable ureter. At the same time, disposable endoscopes can prevent cross infection and provide patients with a safer diagnosis and treatment experience, with immeasurable potential health economic value. Cost breakdown of reusable endoscopic systems
Disposable endoscopes have greater health and economic value. A study pointed out that the cost of a single operation of domestic traditional flexible ureteroscope was more than 12000 yuan, and the cost of daily disinfection and sterilization and operation and maintenance accounted for 43% of the total cost. Disposable endoscopes do not need to consider lifespan or maintainability, resulting in reduced usage costs. Taking into account the equipment and consumables, the initial cost of performing surgery with a disposable ureteroscope is 5% of that of a reusable ureter. At the same time, disposable endoscopes can prevent cross infection and provide patients with a safer diagnosis and treatment experience, with immeasurable potential health economic value. Cost breakdown of reusable endoscopic systems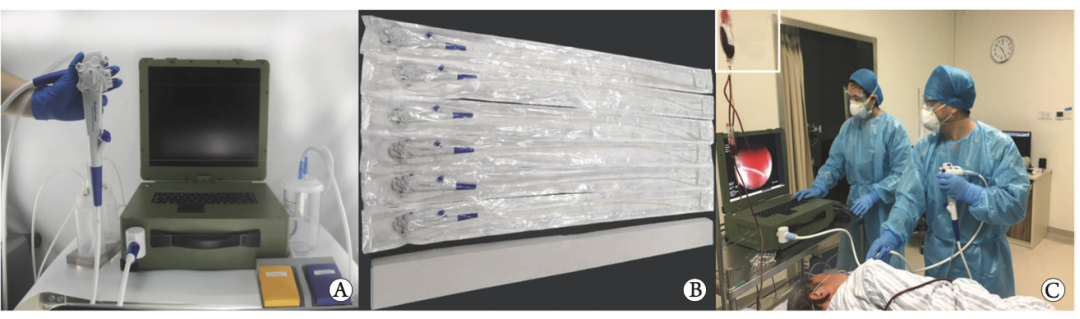 Data source: CNKI, Integrated Research
Data source: CNKI, Integrated Research
 数据来源:融汇
数据来源:融汇
Disposable endoscopes can solve the clinical pain points of cross infection during the use of medical endoscopes. Endoscope is a medical device that comes into close contact with the patient's skin, mucous membranes, and sterile tissues. However, due to the presence of multiple small and long open channels in the endoscope structure, it provides an environment for residual microorganisms, secretions, and blood, as well as cross infection. Medical accidents caused by cross infection often occur. Under the COVID-19 epidemic, the problem of cross infection has attracted great attention. The novel coronavirus can bind to the angiotensin converting enzyme 2 (ACE2) receptor, which exists in the epithelial cells of the lung, upper esophagus, ileum and colon, meaning that the respiratory system and digestive system can bind to the virus. The expert consensus released by the Chinese Medical Association during the epidemic mentioned the use of disposable endoscopes.
 Disposable endoscopes have greater health and economic value. A study pointed out that the cost of a single operation of domestic traditional flexible ureteroscope was more than 12000 yuan, and the cost of daily disinfection and sterilization and operation and maintenance accounted for 43% of the total cost. Disposable endoscopes do not need to consider lifespan or maintainability, resulting in reduced usage costs. Taking into account the equipment and consumables, the initial cost of performing surgery with a disposable ureteroscope is 5% of that of a reusable ureter. At the same time, disposable endoscopes can prevent cross infection and provide patients with a safer diagnosis and treatment experience, with immeasurable potential health economic value. Cost breakdown of reusable endoscopic systems
Disposable endoscopes have greater health and economic value. A study pointed out that the cost of a single operation of domestic traditional flexible ureteroscope was more than 12000 yuan, and the cost of daily disinfection and sterilization and operation and maintenance accounted for 43% of the total cost. Disposable endoscopes do not need to consider lifespan or maintainability, resulting in reduced usage costs. Taking into account the equipment and consumables, the initial cost of performing surgery with a disposable ureteroscope is 5% of that of a reusable ureter. At the same time, disposable endoscopes can prevent cross infection and provide patients with a safer diagnosis and treatment experience, with immeasurable potential health economic value. Cost breakdown of reusable endoscopic systems Data source: CNKI, Integrated Research
Data source: CNKI, Integrated ResearchConsumables lead the innovation of endoscopic products. Reusable endoscopes can easily reach millions, and for hospitals, the longer the service life of equipment, the better, and the stronger the universality of equipment, the better. After the consumables transformation of endoscopes, the cost has rapidly decreased. Endoscopes are no longer expensive equipment, and consumables transformation will lead endoscopes to develop towards "specialization". Product innovation will be carried out around various clinical surgical requirements to improve diagnosis and treatment efficiency: (1) Starting from specific surgical scenarios, the integration innovation of disposable endoscopes will abandon the universal requirements of reusable endoscopes, and design an endoscope specifically designed for specific surgical procedures, such as ERCP dedicated endoscopes, which highly integrate various consumables required for surgery on the endoscope body, no longer requiring doctors to repeatedly operate through forceps. For example, the highly integrated technology of endoscope+ultrasound probe and endoscope+navigation has enormous innovation potential. (2) Portable and flexible, it can expand emergency, bedside, and grassroots diagnosis and treatment scenarios. The lightweight, portable, and non washable disposable endoscope body and mainframe make the application scenarios of endoscopes more flexible, especially suitable for bedside, emergency, grassroots, and other scenarios. In 2020, a domestic study began to explore the application of disposable gastrointestinal endoscopy systems in emergency departments. Some studies have also suggested that the use of portable disposable bronchoscopes at the bedside can detect lesions early and enable timely diagnosis and treatment. Application of portable disposable endoscope in emergency department
 Data source: CNKI, Integrated Research
Data source: CNKI, Integrated Research
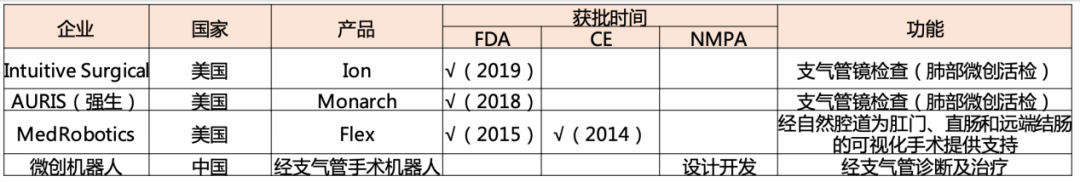
(3) The innovative integration of medical device products and the consumables of endoscopes have reduced the requirement for long-term durability, relying on the development of underlying electronic component technology and the rich demand for clinical scenarios. Disposable endoscopes themselves will inevitably enter a process of rapid iteration and upgrading. As the fundamental platform for endoscopic surgery, endoscopes are also innovatively integrated with other medical equipment products. The combination of disposable endoscope and surgical robot can promote the evolution of flexible surgical robots, and disposable endoscope and ablation intervention will lead a new mode of diagnosis and treatment. Overview of Abdominal Surgery Robots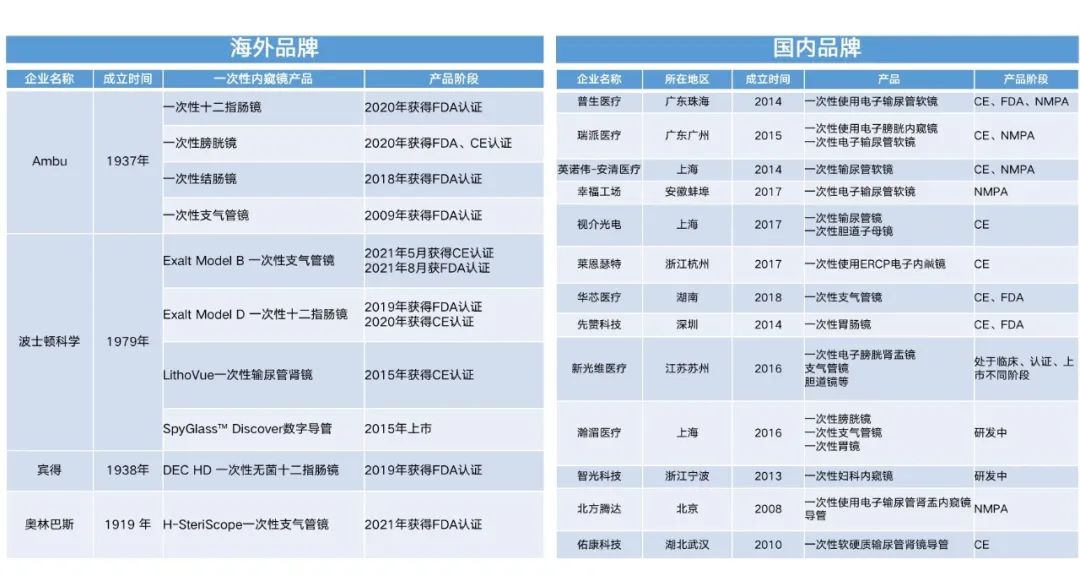 Data source: Southwest Securities, Ronghui Research
Data source: Southwest Securities, Ronghui Research
The huge commercial potential released by the specialized, low-cost, and consumable advantages of disposable endoscopes has attracted the attention of both foreign and domestic enterprises. Unlike traditional endoscopes, disposable endoscopes have significant differences in design concepts, mass production uniformity, and cost control compared to reusable endoscopes. This also makes it difficult for established foreign-funded enterprises to have a clear leading advantage in exploring emerging markets. The disposable bronchoscope products launched by the industry leader Olympus are also agents for domestic brand products. Domestic enterprises have shown a trend of competition, and according to incomplete statistics, there are already 13 disposable endoscope companies. 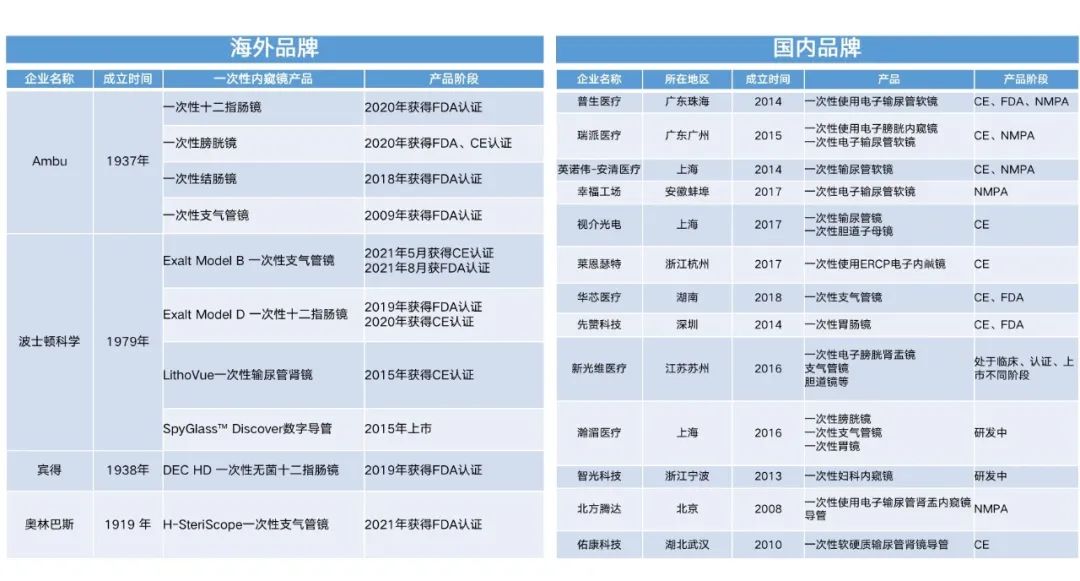
- Here are some representatives of overseas brands:
- Ambu, the world's largest disposable endoscope supplier, was founded in 1937 in Denmark and focuses on three core business areas: anesthesia, patient monitoring and diagnosis, and emergency care. In 2009, Ambu launched the world's first disposable bronchoscope, officially entering the field of disposable endoscopes, and has now developed into the absolute king of disposable bronchoscopy. The main product matrix now includes bronchoscopes, nasopharyngoscopes, duodenoscopes, and cystoscopes. In 2020, Ambu's endoscope sales exceeded 1 million units, with a revenue of 230 million euros. 2. Boston Scientific - Innovative Breakthrough of High Value Consumables Giant Founded in 1979, Boston Scientific is a leading global medical technology company with products covering radiology, cardiology, peripheral, neuromodulation, electrophysiology, endoscopy, and more. BOCO has launched the world's first disposable ureteroscope and disposable duodenoscope, and its product line also includes bronchoscopes, gastroscopes, and cholangioscopes. 3. Olympus - Global King of Soft Mirrors Olympus was founded in 1919, initially focusing on microscope business. Since 1952, the company has been laying out medical endoscope products and has significant technological advantages in image clarity and smooth operation. At present, the company's global market share for soft mirrors has reached 65%, while the global market share for hard mirrors is 22%. In 2019, the company announced its entry into the field of disposable endoscopes. In May 2021, the company launched 5 disposable H-SteriScope bronchoscopes, which can be used for the diagnosis and treatment of lung diseases.
- Here are some representatives of domestic brands:
1. Zhuhai Pusen - the earliest disposable endoscope enterprise in China, Pusen was established in 2014. The company's disposable electronic ureteroscope has obtained FDA and CE certification and has been sold overseas, with markets covering Europe, America, the Middle East, Asia Pacific, Africa, Australia and other regions. The cooperative medical schools include the Mayo Clinic and Cleveland Clinic in the United States. In June 2020, the product was approved by NMPA. The company's product line also includes disposable nasopharyngoscopes, disposable bronchoscopes, disposable cystoscopes, etc. 2. Guangzhou Ruipai - the first enterprise to obtain a Class III registration certificate for disposable electronic bladder endoscopes. Guangzhou Ruipai was established in 2015 and is a global supplier mainly providing integrated solutions for minimally invasive surgery with disposable endoscopes. The company's product layout includes urology, gynecology, otolaryngology, respiratory, and general surgery. The company obtained its first NMPA registration certificate for disposable ureteroscopes and disposable electronic bladder endoscopes in 2020. 3. Xinguang Wei - a domestic alternative to entering the high-end market of endoscopes. Founded in 2016, Xinguang Wei's product matrix covers soft and hard endoscopes, and has 4K imaging technology, 3D imaging technology, special light imaging technology, ultra-fine endoscopic imaging technology, endoscopic consumables technology, etc. Its multiple disposable electronic endoscope products have entered clinical, registration, or sales stages in China, the United States, Europe, and other countries and regions. In August 2021, Xinguang Wei Medical completed nearly 400 million yuan in Series B financing, with investors including Hudson Bay Capital Management, Prime Capital and other new investment institutions, and continued support from old shareholders Hillhouse Capital, Eli Lilly Asia Fund, and Jingwei China.
Conclusion: With the competition among hundreds of boats, those who strive for excellence are the first. The disposable endoscope market has just begun, and we believe that with the maturity of the supply chain system, more manufacturers will enter this market. We believe that in the future, companies with the following abilities are expected to stand out: firstly, companies with independent and continuous research and development capabilities. The rapid development of underlying electronic components such as CMOS and backend image processing chips, as well as the scenario based innovation brought about by consumables, will inevitably promote the rapid upgrading of disposable endoscope products. The second is enterprises with strong supply chain management and cost control capabilities. The current charging system in China restricts the promotion of disposable endoscopes. Only with truly low cost can they be well integrated into the current charging system and truly promote product sales. The third is enterprises with large-scale production and manufacturing capabilities and experience. Unlike the craftsman style assembly production of reusable endoscopes, the significant increase in usage under consumables requires automated production lines and supporting quality control systems.
Source: Arterial network
(3) The innovative integration of medical device products and the consumables of endoscopes have reduced the requirement for long-term durability, relying on the development of underlying electronic component technology and the rich demand for clinical scenarios. Disposable endoscopes themselves will inevitably enter a process of rapid iteration and upgrading. As the fundamental platform for endoscopic surgery, endoscopes are also innovatively integrated with other medical equipment products. The combination of disposable endoscope and surgical robot can promote the evolution of flexible surgical robots, and disposable endoscope and ablation intervention will lead a new mode of diagnosis and treatment. Overview of Abdominal Surgery Robots Data source: Southwest Securities, Ronghui Research
Data source: Southwest Securities, Ronghui Research
The huge commercial potential released by the specialized, low-cost, and consumable advantages of disposable endoscopes has attracted the attention of both foreign and domestic enterprises. Unlike traditional endoscopes, disposable endoscopes have significant differences in design concepts, mass production uniformity, and cost control compared to reusable endoscopes. This also makes it difficult for established foreign-funded enterprises to have a clear leading advantage in exploring emerging markets. The disposable bronchoscope products launched by the industry leader Olympus are also agents for domestic brand products. Domestic enterprises have shown a trend of competition, and according to incomplete statistics, there are already 13 disposable endoscope companies. 
- Here are some representatives of overseas brands:
- Ambu, the world's largest disposable endoscope supplier, was founded in 1937 in Denmark and focuses on three core business areas: anesthesia, patient monitoring and diagnosis, and emergency care. In 2009, Ambu launched the world's first disposable bronchoscope, officially entering the field of disposable endoscopes, and has now developed into the absolute king of disposable bronchoscopy. The main product matrix now includes bronchoscopes, nasopharyngoscopes, duodenoscopes, and cystoscopes. In 2020, Ambu's endoscope sales exceeded 1 million units, with a revenue of 230 million euros. 2. Boston Scientific - Innovative Breakthrough of High Value Consumables Giant Founded in 1979, Boston Scientific is a leading global medical technology company with products covering radiology, cardiology, peripheral, neuromodulation, electrophysiology, endoscopy, and more. BOCO has launched the world's first disposable ureteroscope and disposable duodenoscope, and its product line also includes bronchoscopes, gastroscopes, and cholangioscopes. 3. Olympus - Global King of Soft Mirrors Olympus was founded in 1919, initially focusing on microscope business. Since 1952, the company has been laying out medical endoscope products and has significant technological advantages in image clarity and smooth operation. At present, the company's global market share for soft mirrors has reached 65%, while the global market share for hard mirrors is 22%. In 2019, the company announced its entry into the field of disposable endoscopes. In May 2021, the company launched 5 disposable H-SteriScope bronchoscopes, which can be used for the diagnosis and treatment of lung diseases.
- Here are some representatives of domestic brands:
Conclusion: With the competition among hundreds of boats, those who strive for excellence are the first. The disposable endoscope market has just begun, and we believe that with the maturity of the supply chain system, more manufacturers will enter this market. We believe that in the future, companies with the following abilities are expected to stand out: firstly, companies with independent and continuous research and development capabilities. The rapid development of underlying electronic components such as CMOS and backend image processing chips, as well as the scenario based innovation brought about by consumables, will inevitably promote the rapid upgrading of disposable endoscope products. The second is enterprises with strong supply chain management and cost control capabilities. The current charging system in China restricts the promotion of disposable endoscopes. Only with truly low cost can they be well integrated into the current charging system and truly promote product sales. The third is enterprises with large-scale production and manufacturing capabilities and experience. Unlike the craftsman style assembly production of reusable endoscopes, the significant increase in usage under consumables requires automated production lines and supporting quality control systems.
Source: Arterial network









